Adverse Events Following Immunisation (AEFI) refer to any untoward medical occurrence after vaccination, which may or may not have a causal relationship with the vaccine, as defined by the World Health Organization. Monitoring AEFI is a critical component of immunisation programmes because vaccines are administered to healthy populations, especially children and adolescents, making safety assurance essential to sustain public trust.
This assumes particular importance in India, where cervical cancer accounts for over 127,000 new cases and nearly 80,000 deaths annually, making the introduction of nationwide Human Papillomavirus (HPV) vaccination a major preventive intervention. At the same time, the resurgence of measles outbreaks across multiple states in the United States, driven partly by vaccine hesitancy, highlights how inadequate trust in vaccine safety can reverse decades of public health gains.
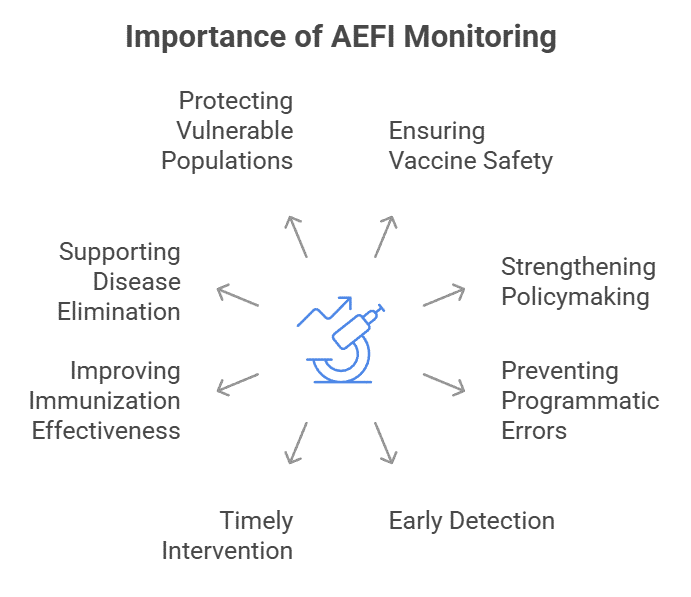
I. Public Health and Scientific Importance of AEFI Monitoring
1. Ensuring Vaccine Safety and Risk–Benefit Balance
- Scientific validation of vaccine safety: Continuous AEFI monitoring allows identification of rare adverse reactions, ensuring that the benefits of vaccination outweigh risks, which is essential since vaccines are given to millions of healthy individuals.
- Strengthening evidence-based policymaking: Surveillance helps governments refine vaccination schedules, dosage, and eligibility criteria based on real-world safety data.
- Example: The adoption of a single-dose HPV vaccine schedule, recommended globally, was supported by safety and efficacy data accumulated through AEFI surveillance.
- Preventing programmatic errors and improving delivery: AEFI monitoring can identify errors related to storage, transport, or administration rather than the vaccine itself.
2. Early Detection and Rapid Response to Safety Signals
- Identification of rare or unexpected adverse events: Even clinical trials cannot detect extremely rare side effects; post-marketing surveillance helps identify such occurrences.
- Timely intervention and corrective action: Early detection allows authorities to suspend batches, modify protocols, or strengthen training if necessary.
- Example: India’s COVID-19 vaccination programme included real-time AEFI reporting, enabling rapid investigation of adverse cases and reinforcing programme safety.
- Strengthening pharmacovigilance ecosystem: Continuous surveillance contributes to long-term scientific understanding of vaccine safety profiles.
3. Improving Immunisation Effectiveness and Health Outcomes
- Maintaining high immunisation coverage: Public confidence in vaccine safety encourages widespread participation, which is essential for achieving herd immunity.
- Example: Countries implementing widespread HPV vaccination have demonstrated significant reductions in HPV infections, precancerous lesions, and cervical cancer incidence.
- Supporting disease elimination goals: Safe vaccination programmes help eliminate vaccine-preventable diseases.
- Protecting vulnerable populations: Women, children, and immunocompromised individuals benefit significantly from vaccines when safety is assured.
II. Role of AEFI Monitoring in Sustaining Public Trust and Combating Vaccine Hesitancy
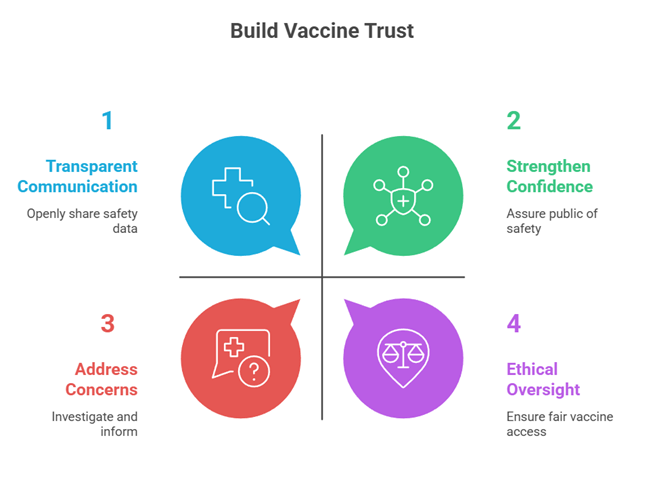
1. Ensuring Transparency and Accountability
- Building credibility of public health institutions: Transparent investigation and reporting of AEFI enhance trust in health authorities.
- Preventing misinformation and speculation: Open communication prevents rumours that vaccines are unsafe.
- Example: Transparent communication during COVID-19 vaccination helped counter false claims about severe adverse reactions.
- Strengthening democratic accountability: Public institutions must justify interventions affecting millions of citizens.
2. Countering Vaccine Hesitancy and Anti-Vaccination Sentiment
- Preventing decline in vaccination rates: Vaccine hesitancy can reduce coverage and lead to disease resurgence.
- Example: CASE STUDY – Measles Resurgence in the United States: Declining vaccination due to safety fears contributed to outbreaks across multiple states.
- Reinforcing scientific trust in preventive healthcare: Demonstrating strong safety systems reassures citizens about vaccine reliability.
- Preventing erosion of herd immunity: Declining immunisation rates weaken community protection.
3. Ethical Responsibility and Social Legitimacy
- Protecting individual rights and public welfare: Monitoring ensures ethical administration of vaccines with minimal harm.
- Example: Ethical vaccination programmes include informed consent and post-vaccination observation.
- Ensuring equity and justice in public health: Marginalized populations must receive safe vaccines without disproportionate risks.
- Promoting social acceptance of new vaccines: New vaccine introductions require strong trust mechanisms.
- Example: CASE STUDY – Introduction of Cervavac, developed by Serum Institute of India, requires transparent safety monitoring to ensure acceptance.
III. Institutional and Governance Dimensions: India’s Framework and Challenges
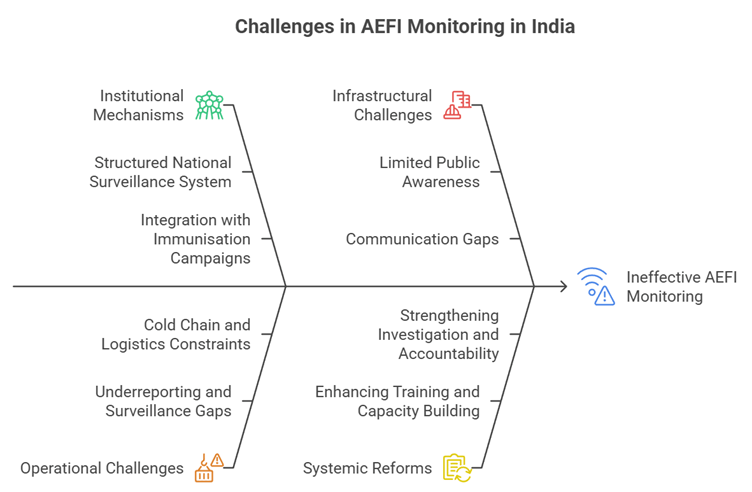
1. Existing Institutional Mechanisms for AEFI Monitoring
- Structured national surveillance system: India has a multi-tier AEFI monitoring mechanism involving national, state, and district committees.
- Integration with immunisation campaigns: Large-scale vaccination programmes incorporate safety monitoring as a core component.
- Use of digital technologies: Digital platforms enable real-time reporting and tracking of adverse events.
- Example: COVID-19 vaccination platforms enabled efficient monitoring and follow-up.
2. Operational and Infrastructural Challenges
- Underreporting and surveillance gaps: Rural and remote areas may lack trained personnel and reporting systems.
- Cold chain and logistics constraints: Improper storage conditions can compromise vaccine safety and effectiveness.
- Limited public awareness and communication gaps: Lack of awareness may cause fear or misinterpretation of adverse events.
- Example: Misunderstanding minor side effects can reduce vaccine acceptance.
3. Need for Strengthening Surveillance and Systemic Reforms
- Enhancing training and capacity building: Healthcare workers need specialized training to detect and manage AEFI.
- Strengthening investigation and accountability mechanisms: Independent investigation enhances transparency and credibility.
- Example: Scientific investigation of vaccine safety builds trust in public institutions.
- Improving community engagement and communication: Public awareness campaigns help explain risks and benefits clearly.
Conclusion:
- Monitoring Adverse Events Following Immunisation is indispensable for ensuring vaccine safety, sustaining public trust, and achieving public health goals. With India accounting for nearly one-fifth of global cervical cancer burden, HPV vaccination represents a transformative preventive intervention. However, its success depends heavily on transparent reporting, robust surveillance, effective cold chain management, and community engagement.
- Strengthening pharmacovigilance, enhancing institutional capacity, and ensuring accountability can help India achieve global targets such as the WHO’s cervical cancer elimination strategy, which aims for 90% vaccination coverage among adolescent girls.
- A transparent and scientifically robust AEFI monitoring system will not only safeguard individual health but also ensure long-term societal confidence in immunisation as one of the most effective tools of preventive healthcare.